Myasthenia gravis
What is myasthenia gravis?
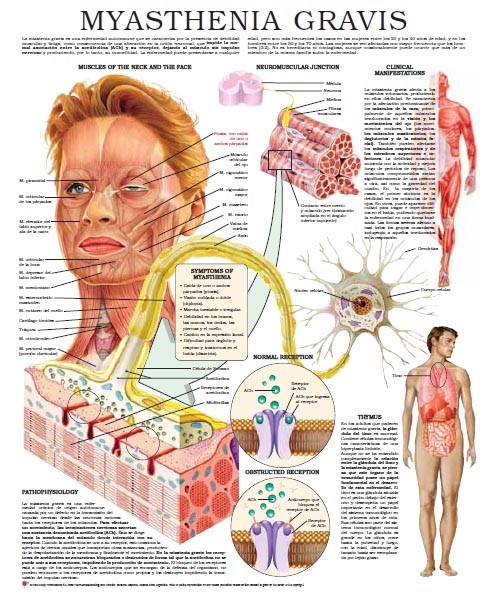
Myasthenia gravis is a chronic autoimmune neuromuscular disease that causes weakness in the skeletal muscles, which are responsible for breathing and moving parts of the body, including the arms and legs. The name myasthenia gravis, which is Latin and Greek in origin, means “grave, or serious, muscle weakness.”
The hallmark of myasthenia gravis is muscle weakness that worsens after periods of activity and improves after periods of rest. Certain muscles such as those that control eye and eyelid movement, facial expression, chewing, talking, and swallowing are often (but not always) involved in the disorder. The muscles that control breathing and neck and limb movements may also be affected.
There is no known cure but with current therapies most cases of myasthenia gravis are not as “grave” as the name implies. Available treatments can control symptoms and often allow people to have a relatively high quality of life. Most individuals with the condition have a normal life expectancy.
What causes myasthenia gravis?
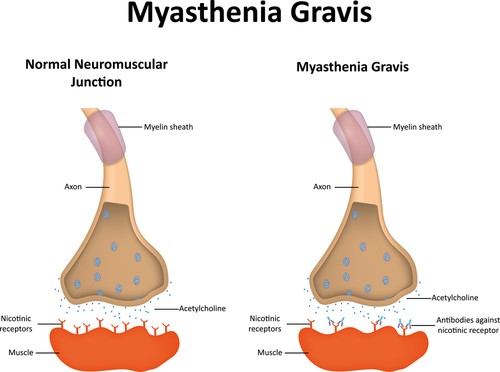
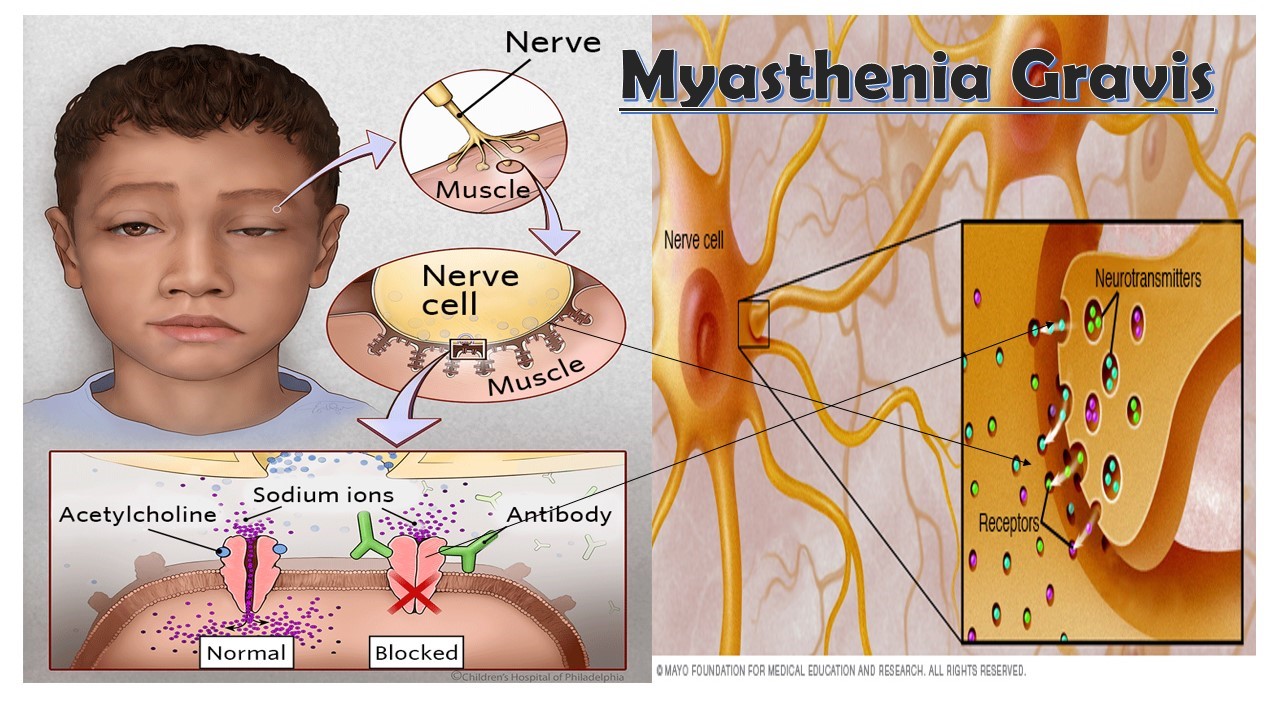
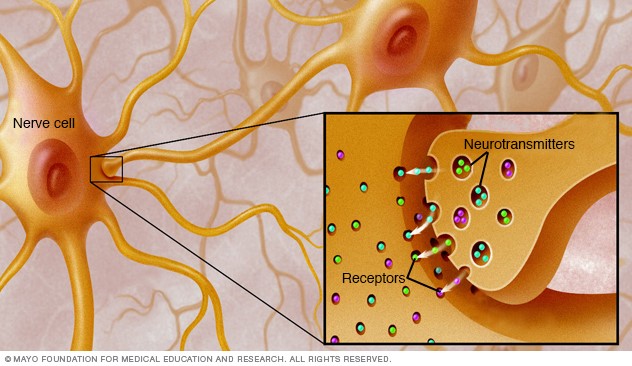
Myasthenia gravis is caused by an error in the transmission of nerve impulses to muscles. It occurs when normal communication between the nerve and muscle is interrupted at the neuromuscular junction—the place where nerve cells connect with the muscles they control.
Neurotransmitters are chemicals that neurons, or brain cells, use to communicate information. Normally when electrical signals or impulses travel down a motor nerve, the nerve endings release a neurotransmitter called acetylcholine. Acetylcholine travels from the nerve ending and binds to acetylcholine receptors on the muscle. The binding of acetylcholine to its receptor activates the muscle and causes a muscle contraction.
In myasthenia gravis, antibodies (immune proteins) block, alter, or destroy the receptors for acetylcholine at the neuromuscular junction, which prevents the muscle from contracting. In most individuals with myasthenia gravis, this is caused by antibodies to the acetylcholine receptor itself. However, antibodies to other proteins, such as MuSK (Muscle-Specific Kinase) protein, can also lead to impaired transmission at the neuromuscular junction.
These antibodies are produced by the body’s own immune system. Myasthenia gravis is an autoimmune disease because the immune system—which normally protects the body from foreign organisms—mistakenly attacks itself.
The thymus is a gland that controls immune function and maybe associated with myasthenia gravis. Located in the chest behind the breast bone, the gland is largest in children. It grows gradually until puberty, and then gets smaller and is replaced by fat. Throughout childhood, the thymus plays an important role in the development of the immune system because it is responsible for producing T-lymphocytes or T cells, a specific type of white blood cell that protects the body from viruses and infections.
In many adults with myasthenia gravis, the thymus gland remains large. People with the disease typically have clusters of immune cells in their thymus gland similar to lymphoid hyperplasia—a condition that usually only happens in the spleen and lymph nodes during an active immune response. Some individuals with myasthenia gravis develop thymomas (tumors of the thymus gland). Thymomas are most often harmless, but they can become cancerous.
The thymus gland plays a role in myasthenia gravis, but its function is not fully understood. Scientists believe that the thymus gland may give incorrect instructions to developing immune cells, ultimately causing the immune system to attack its own cells and tissues and produce acetylcholine receptor antibodies—setting the stage for the attack on neuromuscular transmission.
What are the symptoms of myasthenia gravis?
Although myasthenia gravis may affect any skeletal muscle, muscles that control eye and eyelid movement, facial expression, and swallowing are most frequently affected. The onset of the disorder may be sudden and symptoms often are not immediately recognized as myasthenia gravis.
In most cases, the first noticeable symptom is weakness of the eye muscles. In others, difficulty swallowing and slurred speech may be the first signs. The degree of muscle weakness involved in myasthenia gravis varies greatly among individuals, ranging from a localized form limited to eye muscles (ocular myasthenia), to a severe or generalized form in which many muscles—sometimes including those that control breathing—are affected.
Symptoms may include:
• drooping of one or both eyelids (ptosis)
• blurred or double vision (diplopia) due to weakness of the muscles that control eye movements
• a change in facial expression
• difficulty swallowing
• shortness of breath
• impaired speech (dysarthria)
• weakness in the arms, hands, fingers, legs, and neck.
Who gets myasthenia gravis?
Myasthenia gravis affects both men and women and occurs across all racial and ethnic groups. It most commonly impacts young adult women (under 40) and older men (over 60), but it can occur at any age, including childhood. Myasthenia gravis is not inherited nor is it contagious. Occasionally, the disease may occur in more than one member of the same family.
Although myasthenia gravis is rarely seen in infants, the fetus may acquire antibodies from a mother affected with myasthenia gravis—a condition called neonatal myasthenia. Generally, neonatal myasthenia gravis is temporary and the child’s symptoms usually disappear within two to three months after birth. Rarely, children of a healthy mother may develop congenital myasthenia. This is not an autoimmune disorder (it is caused by defective genes that produce abnormal proteins in the neuromuscular junction) and can cause similar symptoms to myasthenia gravis.
How is myasthenia gravis diagnosed?
A doctor may perform or order several tests to confirm the diagnosis, including:
• A physical and neurological examination. A physician will first review an individual’s medical history and conduct a physical examination. In a neurological examination, the physician will check muscle strength and tone, coordination, sense of touch, and look for impairment of eye movements.
• An edrophonium test. This test uses injections of edrophonium chloride to briefly relieve weakness in people with myasthenia gravis. The drug blocks the breakdown of acetylcholine and temporarily increases the levels of acetylcholine at the neuromuscular junction. It is usually used to test ocular muscle weakness.
• A blood test. Most individuals with myasthenia gravis have abnormally elevated levels of acetylcholine receptor antibodies. A second antibody—called the anti-MuSK antibody—has been found in about half of individuals with myasthenia gravis who do not have acetylcholine receptor antibodies. A blood test can also detect this antibody. However, in some individuals with myasthenia gravis, neither of these antibodies is present. These individuals are said to have seronegative (negative antibody) myasthenia.
• Electrodiagnostics. Diagnostic tests include repetitive nerve stimulation, which repeatedly stimulates a person’s nerves with small pulses of electricity to tire specific muscles. Muscle fibers in myasthenia gravis, as well as other neuromuscular disorders, do not respond as well to repeated electrical stimulation compared to muscles from normal individuals. Single fiber electromyography (EMG), considered the most sensitive test for myasthenia gravis, detects impaired nerve-to-muscle transmission. EMG can be very helpful in diagnosing mild cases of myasthenia gravis when other tests fail to demonstrate abnormalities.
• Diagnostic imaging. Diagnostic imaging of the chest using computed tomography (CT) or magnetic resonance imaging (MRI) may identify the presence of a thymoma.
• Pulmonary function testing. Measuring breathing strength can help predict if respiration may fail and lead to a myasthenic crisis.
Because weakness is a common symptom of many other disorders, the diagnosis of myasthenia gravis is often missed or delayed (sometimes up to two years) in people who experience mild weakness or in those individuals whose weakness is restricted to only a few muscles.
What is a myasthenic crisis?
A myasthenic crisis is a medical emergency that occurs when the muscles that control breathing weaken to the point where individuals require a ventilator to help them breathe.
Approximately 15 to 20 percent of people with myasthenia gravis experience at least one myasthenic crisis. This condition usually requires immediate medical attention and may be triggered by infection, stress, surgery, or an adverse reaction to medication. However, up to one-half of people may have no obvious cause for their myasthenic crisis. Certain medications have been shown to cause myasthenia gravis. However, sometimes these medications may still be used if it is more important to treat an underlying condition.
How is myasthenia gravis treated?
Today, myasthenia gravis can generally be controlled. There are several therapies available to help reduce and improve muscle weakness.
• Thymectomy. This operation to remove the thymus gland (which often is abnormal in individuals with myasthenia gravis) can reduce symptoms and may cure some people, possibly by rebalancing the immune system. A recent NINDS-funded study found that thymectomy is beneficial both for people with thymoma and those with no evidence of the tumors. The clinical trial followed 126 people with myasthenia gravis and no visible thymoma and found that the surgery reduced muscle weakness and the need for immunosuppressive drugs.
• Anticholinesterase medications. Medications to treat the disorder include anticholinesterase agents such as mestinon or pyridostigmine, which slow the breakdown of acetylcholine at the neuromuscular junction and thereby improve neuromuscular transmission and increase muscle strength.
• Immunosuppressive drugs. These drugs improve muscle strength by suppressing the production of abnormal antibodies. They include prednisone, azathioprine, mycophenolate mofetil, tacrolimus, and rituximab. The drugs can cause significant side effects and must be carefully monitored by a physician.
• Plasmapheresis and intravenous immunoglobulin. These therapies may be options in severe cases of myasthenia gravis. Individuals can have antibodies in their plasma (a liquid component in blood) that attack the neuromuscular junction. These treatments remove the destructive antibodies, although their effectiveness usually only lasts for a few weeks to months.
o Plasmapheresis is a procedure using a machine to remove harmful antibodies in plasma and replace them with good plasma or a plasma substitute.
o Intravenous immunoglobulin is a highly concentrated injection of antibodies pooled from many healthy donors that temporarily changes the way the immune system operates. It works by binding to the antibodies that cause myasthenia gravis and removing them from circulation.
What is the prognosis?
With treatment, most individuals with myasthenia can significantly improve their muscle weakness and lead normal or nearly normal lives. Sometimes the severe weakness of myasthenia gravis may cause respiratory failure, which requires immediate emergency medical care.
Some cases of myasthenia gravis may go into remission—either temporarily or permanently—and muscle weakness may disappear completely so that medications can be discontinued. Stable, long-lasting complete remissions are the goal of thymectomy and may occur in about 50 percent of individuals who undergo this procedure.
What research is being done?
The mission of the National Institute of Neurological Disorders and Stroke (NINDS) is to seek fundamental knowledge about the brain and nervous system and to use that knowledge to reduce the burden of neurological disease. The NINDS is a component of the National Institutes of Health (NIH), the leading supporter of biomedical research in the world.
Although there is no cure for myasthenia gravis, management of the disorder has improved over the past 30 years. There is a greater understanding about the structure and function of the neuromuscular junction, the fundamental aspects of the thymus gland and of autoimmunity, and the disorder itself. Technological advances have led to more timely and accurate diagnosis of myasthenia gravis and new and enhanced therapies have improved treatment options. Researchers are working to develop better medications, identify new ways to diagnose and treat individuals, and improve treatment options.
Medication
Some people with myasthenia gravis do not respond favorably to available treatment options, which usually include long-term suppression of the immune system. New drugs are being tested, either alone or in combination with existing drug therapies, to see if they are effective in treating the disease.
Studies are investigating the use of therapy targeting the B cells that make antibodies (rituximab) or the process by which acetylcholine antibodies injure the neuromuscular junction (eculizumab). The drugs have shown promise in initial clinical trials.
Diagnostics and biomarkers
In addition to developing new medications, researchers are trying to find better ways to diagnose and treat this disorder. For example, NINDS-funded researchers are exploring the assembly and function of connections between nerves and muscle fibers to understand the fundamental processes in neuromuscular development. This research could reveal new therapies for neuromuscular diseases like myasthenia gravis.
Researchers are also exploring better ways to treat myasthenia gravis by developing new tools to diagnose people with undetectable antibodies and identify potential biomarkers (signs that can help diagnose or measure the progression of a disease) to predict an individual’s response to immunosuppressive drugs.
New treatment options
Findings from a recent NINDS-supported study yielded conclusive evidence about the benefits of surgery for individuals without thymoma, a subject that had been debated for decades. Researchers hope that this trial will become a model for rigorously testing other treatment options, and that other studies will continue to examine different therapies to see if they are superior to standard care options.